
This rule was discovered in the year 1925 by Friedrich Hund. It is a rule which depends on the observation of atomic spectra, which is helpful in predicting the ground state of a molecule or an atom with one or more than one open electronic shells. It says if two or more than two orbitals having the same amount of energy are unoccupied then the electrons will start occupying them individually before they fill them in pairs. The rule states that, for a stated electron configuration, the greatest value of spin multiplicity has the lowest energy term. These type of elements do not react with other elements. The orbitals that have full energy level are the most stable, for example, noble gases. When all the orbitals of an atom are full it is most stable. The stability can also be predicted by the electron configuration.  
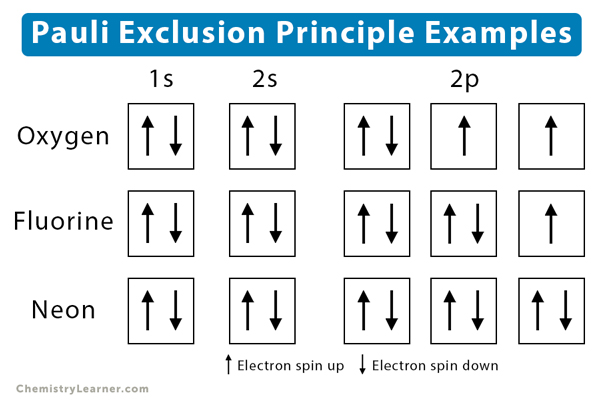
Similar chemical characteristics can be seen in elements that have similar valence numbers. The chemical characteristics of an element are largely dependent on the valence electrons. When valence shells are not full then the atom is the least stable. The valence shells of two atoms that come in contact with each other will interact first. The above image helps in understanding the electronic configuration and its purpose. The same orbital will be occupied by the two 2s electrons although different orbitals will be occupied by the two 2p electrons in reference to Hund’s rule. For instance, a carbon atom’s electron configuration would be 1s 22s 22p 2. The initial electrons spin in the sub-level decides what the spin of the other electrons would be. When we consider the second rule, the spins of unpaired electrons in singly occupied orbitals are the same. The electrons do not share orbitals to reduce repulsion. The electrons repel each other as they are negatively charged. The electrons enter an empty orbital before pairing up.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
